Tissue plasminogen activator (tPA, Alteplase) catalyzes the conversion of plasminogen to plasmin, the major enzyme responsible for clot breakdown. Its fibrinolysis properties makes it clinically useful for treatment of treatment of myocardial infarction with ST-elevation (STEMI), acute ischemic emboilc or thrombotic stroke (AIS), acute massive pulmonary embolism, and central venous access devices (CVAD). Alteplase is produced by Genentech, and is manufactured using recombinant biotechnology techniques. Other r-tPA medications are Reteplase and Tenecteplase.
Unfortunately these medications are not used daily, and often only in emergent situations. For facilities without 24-hour pharmacy coverage, it is often left to the nursing staff or physician to prepare the medication. The preparation instructions included in the kit are complicated and long, which make it difficult to go through during stressful situations. It is beneficial for you to be familiar with the procedure before the need arises. In this tutorial I will walk you through the mixing procedure.
Step 1
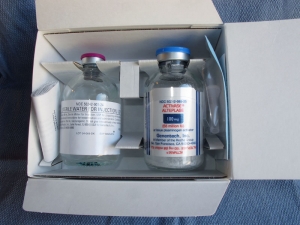
When you the open the kit you will find a vial of sterile water and a sterile vial of powdered medication (blue cap).
Notice how the tPA is in powder form.
In the box you will also find the package insert (which is worth reading when you have a chance) and a dual sided spike which is wrapped in sterile packaging. The spike is the same on both sides, and has a dual channel so that air and fluid can flow in opposite directions.
Step 2
Remove the cap of the water vial sterilely. Unwrap the spike and hold it under sterile condiions. Holding the vial in the upright position, spike the vial.
Step 3
Continuing under sterile conditions, remove the cap from the powdered tPA vial. Hold the water vial firmly in the upright position. With your other hand hold the tPA upside down, and lower the vial down onto the spike.
With both vials firmly spiked, invert both vials together. At this point, the water will on top and will flow from the water vial into the tPA vial. Hold the vials firmly, because the high center of gravity will make them unsteady. This process will take about 1 minute. Do not agitate the vials during this process.
Step 4
One all of the water is in the tPA vial, grasp the flanges and decannulate the tPA vial by lifting up on the spike and water vial simultaneously.
Step 5
Gentle swirling will thoroughly mix the contents of the tPA vial. When mixing DO NOT SHAKE THE VIAL. It will produce a nearly unusable foam. Swirl gently. The medication is now ready. It can be drawn up and administered based on the indication and dosing regimen. Be cautious drawing up your dose to administer. If you draw out a dose with a needle and syringe and tip the vial of tPA upside down while doing so, the hole in the stopper left by the large spike will often lead to leakage all over you and/or the floor. This is a pricey leak. Be sure to dispose your sharps.






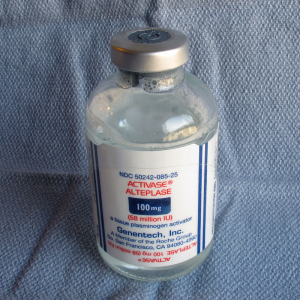

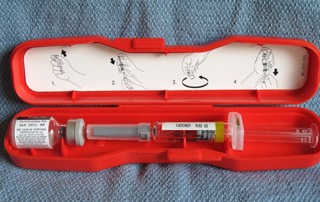
Please note, the diluent is NOT normal saline, but Sterile Water for Injection.
Thank you for the excellent catch of my mistake. The dilutent is absoluetly sterile water. I have updated the article. This is the way #FOAM peer review should work.
It is perfectly fine to also mix it with normal saline
Also be cautious, if you draw out a dose with a needle and syringe and tip the vial of tPA upside down while doing so, the hole in the stopper left by the large spike will often lead to leakage all over you and/or the floor. This is a pricey leak. Also when mixing DO NOT SHAKE THE VIAL. It will produce a nearly unusable foam. Swirl gently.
Thank you for pointing out some excellent additional tips.
[…] tPA Mixing Tutorial – Pay attention here, simple steps to get the drug mixed quickly. […]
[…] Resus Review Blog by Charles Bruen (See the tPA Mixing Tutorial) […]
[…] The Resus Review wrote up tPA mixing instructions […]
Hi. New to the site. At a new hospital (ER RN). Just performed my first tPA here, I have always mixed the lytic (stirred, not shaken) and then inserted my IV tubing, then drawn the waste out with a syringe at the highest y-site. I was told by the stroke coordinator here that TJC states we are to insert a needle into the bottle (as you describe) and pull the waste directly from the bottle, inserting and wasting whilst the fluid is dependent. I came from a center of excellence for stroke and now I am confused. tPA is now the only drug I have ever had to violate all the admixture protocols impressed upon me by pharmacy. I would love to know why? Thanks so much!!