This 45-year-old woman with a medical history which includes medication-controlled essential hypertension, stage 2 chronic kidney disease, type 2 diabetes mellitus, and a 1 pack-per-day cigarette smoking habit presented to the emergency department with <60 minutes of acute onset of severe shortness of breath that awoke her from sleep. She had felt well the previous day and went to bed with no complaints. Around 4 am she was awoken from sleep very dyspneic, with moderate chest “discomfort” over her left chest and radiated to her back, and was unchanged by position or respirations. She denied any other symptoms such as fever, cough, nausea/vomiting, numbness, or abdominal pain.
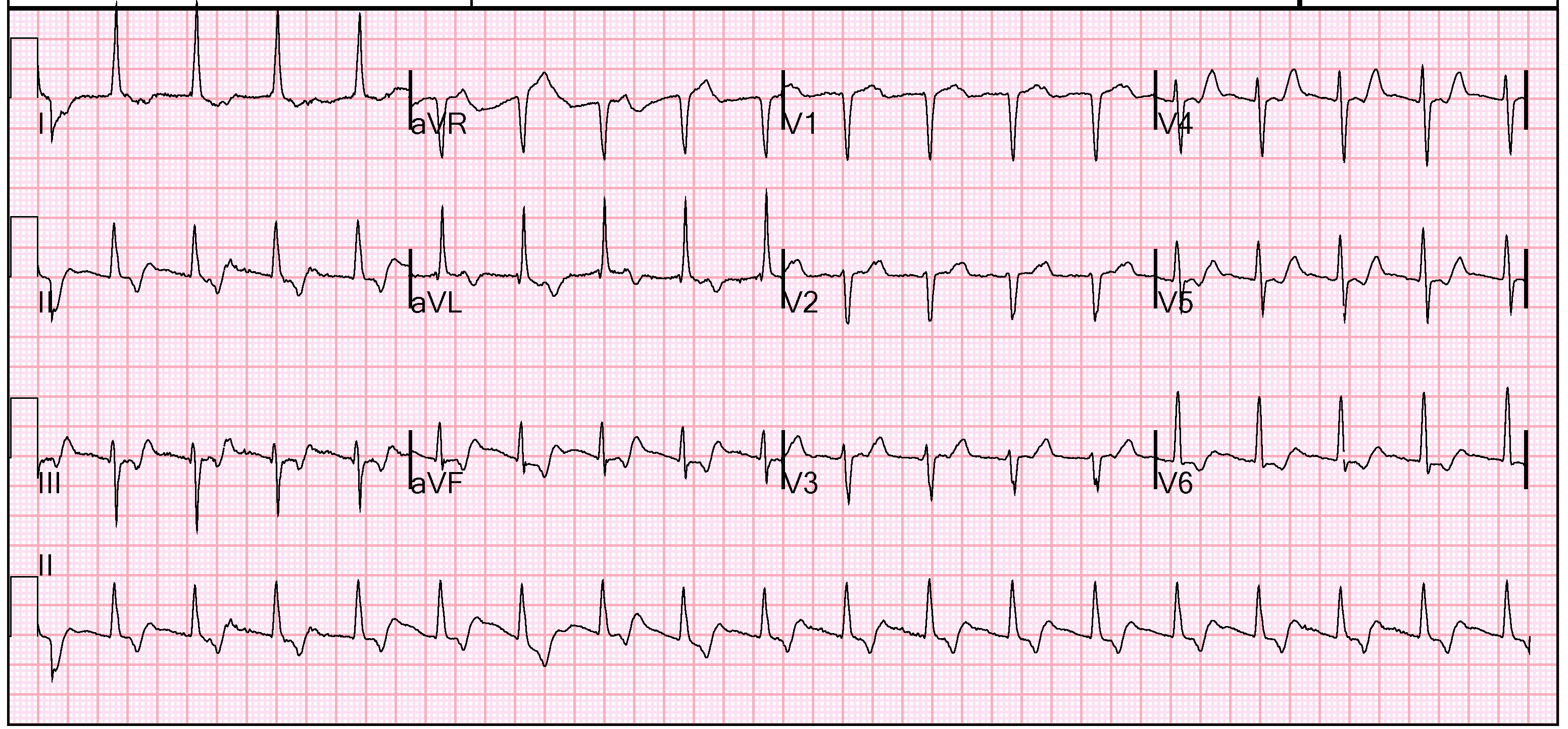
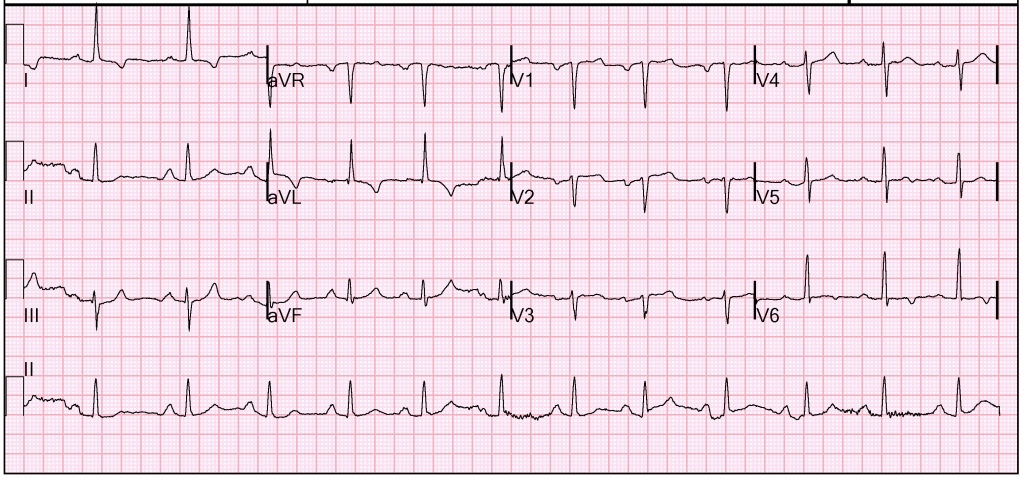
Initial vital signs were blood pressure 138/76 mmHg, pulse 87 bpm, respiratory rate 26, SpO2 was 95%. She was afebrile. Her physical exam was unremarkable; there were clear breath sounds, no S3 or murmur, and no lower extremity edema. She felt slightly less dyspneic with supplemental oxygen. She was treated with an aspirin orally. A sublingual nitroglycerin did improve her chest discomfort. Her chest radiograph was clear. Her presenting electrocardiogram is shown in below.
The patient’s history, presenting complaint, and initial evaluation helps us narrow our differential diagnosis (see Table 1). Waking from sleep with dyspnea, referred to as paroxysmal nocturnal dyspnea, is most commonly associated with congestive heart failure. However, in this patient there is little other evidence of this. She has no history of previous heart disease, her lungs are clear both on exam and imaging which would be the case if there was cardiogenic pulmonary edema, and she does not have lower extremity edema that should develop with vascular congestion. Pneumonia, likewise, is unlikely in a patient without signs/symptoms of infection and normal lung findings. Spontaneous pneumothorax is essentially excluded by the normal radiograph. Obstructive airway disease such as asthma or COPD is an important consideration. The patient has no documented history of either, but her smoking suggests the possibility. Wheezes were not auscultated on physical exam, but a trial of nebulization is appropriate during the workup. Anemia is unlikely given the rapid onset, but measurement of blood counts is reasonable. She has a low pretest probability for pulmonary embolism using the Well’s Criteria, and meets all 8 criteria of the PERC Rule for Pulmonary Embolism (see Table 2), which gives her <2% risk of pulmonary embolism.
| Differential of nocturnal dyspnea |
| Pulmonary embolism |
| Pneumonia |
| Obstructive airway disease |
| Pneumothorax |
| Congestive heart failure |
| Myocardial ischemia |
| Arrhythmia |
| Anemia |
| Gastroesophageal reflux disease |
| Sleep apnea |
| Panic attack/Anxiety |
| PERC Rule for Pulmonary Embolism |
| Age <50 |
| Heart rate <100 bpm |
| SpO2 >94% |
| No prior history of DVT/PE |
| No hemoptysis |
| No exogenous estrogen |
| No clinic signs suggesting DVT |
| Gastroesophageal reflux disease |
| Reference: Kline et al. J Thromb Haemost 2006;6:772 |
The emergency physicians attending to this patient considered myocardial ischemia as the leading diagnosis. They appropriately treated her with supplemental oxygen and a full-dose aspirin 325 mg orally. The presenting ECG was noted to have ST-segment depression in V5 V6, III, and aVF. There is also down-sloping ST-segments in multiple leads. These findings would be consistent with subendocardial ischemia.
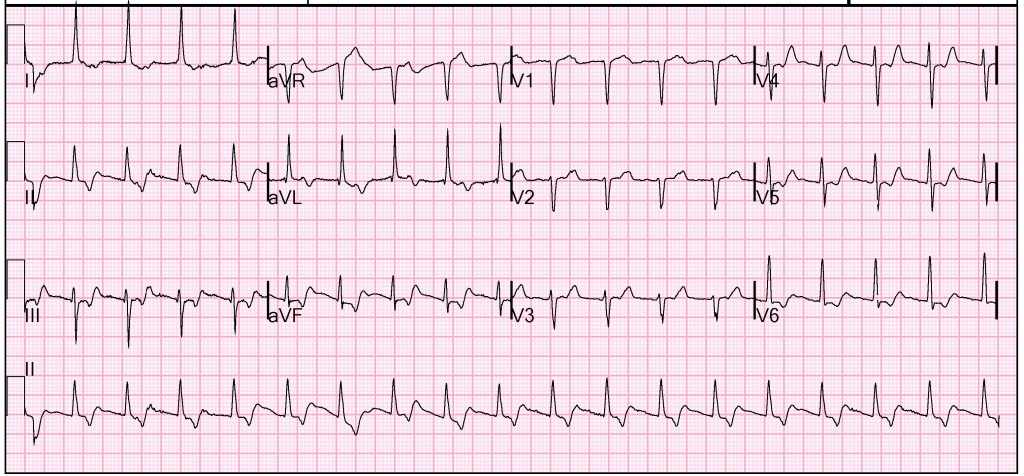
For comparison, the figure below shows an ECG obtained 6 months prior when the patient was not experiencing any symptoms.
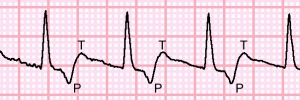
The changes noted between baseline ECG and her presenting ECG would be further concern that the patient was experiencing ischemic chest pain. However, it is always important to fully evaluate all the parts of and ECG. Take a close look at lead II, shown in close-up below in Figure 3. One striking feature is the lack of P-waves preceding the QRS complex. Following the QRS complex however, there is an initial negative deflection and then positive deflection of the tracing. This is actually a retrograde P-wave followed by a normal axis T-wave. This retrograde P-wave is generated from the AV node, and conducts up and to the right depolarizing the atria. From the AV node, the ventricular depolarization and repolarization conducts normally, and can be seen in the normally aligned QRS complex and T-wave. This is classic for an AVNRT rhythm.
Atrioventricular nodal reentrant tachycardia (AVNRT) is the most common form of paroxysmal supraventricular tachycardia (PSVT). Palpitations and lightheadedness are frequent presenting complaints, but in patients with coronary artery disease or structural heart disease chest pain and dyspnea can occur. There is often no specific inciting event for the initiation of the arrhythmia, but alcohol, stimulants, or excessive vagal tone can increase the risk.
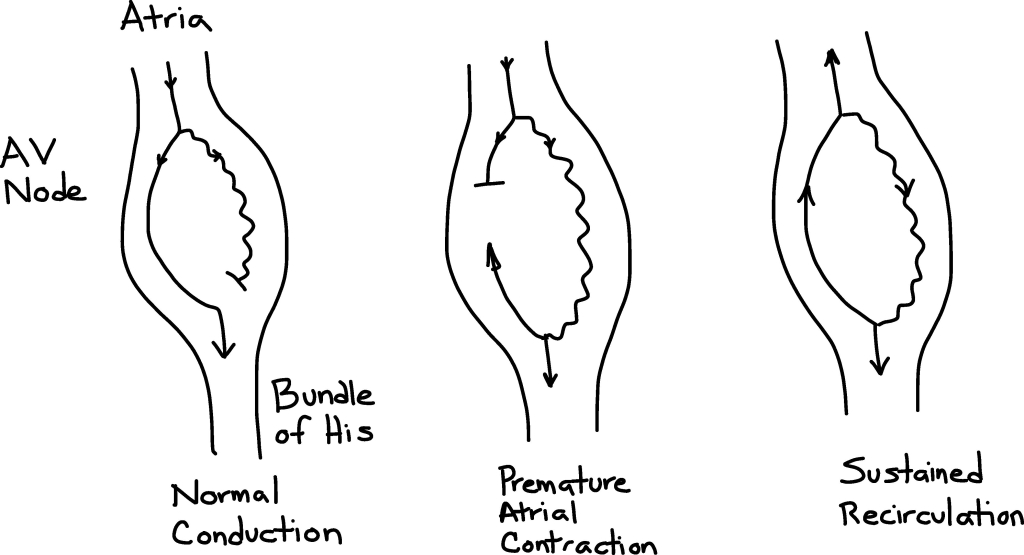
AVNRT arises because there are fast and low conduction pathways through the AV node (see Figure 4). A normal beat is conducted through the fast pathway, and the slow pathway is blocked because of the refractory period in the distal AV node. If a premature atrial contraction occurs (which is the usual inciting mechanism), the fast pathway is refractory, but the slow pathway which while conducting slower has a shorter refractory period and would have recovered excitability and be able to conduct the premature beat. This initial premature beat would have a longer than normal PR interval if captured on ECG. Once the conduction down the slow pathway reaches the common point, it will conduct down the Bundle of His to the ventricle and retrograde up the fast pathway to the atria. The circuit can become self sustaining if the slow pathway has recovered from its refractory period by the time the beat has reached the top of the AV node. There are various forms of AVNRT, but 80-90% of the cases have what is labelled typical AVNRT (slow-fast) and have retrograde conduction via the fast pathway. In this situation, the P-wave is hidden within the QRS complex or seen as an R’ wave. Atypical AVNRT occurs in the opposite direction (fast-slow) with retrograde conduction via the slow pathway and therefore the P-wave can be seen in the ST-segment or as part of the T-wave. Significant ST-segment depression can occur during AVNRT, and represents repolarization changes rather than myocardial ischemia.
Treating the rhythm needs to be divided into acute therapy and prevention of recurrence. As with all arrhythmias causing hemodynamic compromise, electrical cardioversion would be appropriate. If the patient is relatively stable, treatment with adenosine is first-line therapy. Adenosine, a nucleoside analog, has many effects but most important here is the ability to prolong the conduction time through of the AV node, interrupting the re-entry pathways. It is usually given in 6 mg or 12 mg doses. Unfortunately, adenosine is unstable in vivo and is very rapidly metabolized (<10 seconds) by red blood cells and endothelium. Therefore it is administered by fast push. A typical setup is to have a peripheral IV with running 0.9% saline, and inject the adenosine and quickly switch syringes to perform the flush. Alternatively, the adenosine can be combined in a single syringe with 20 ml of 0.9% saline and pushed together all at once.
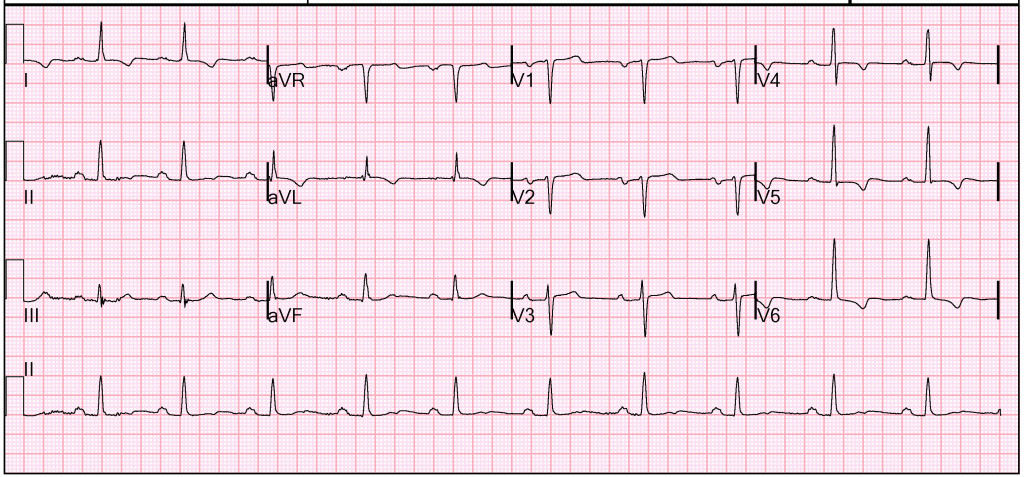
For this patient, her ECG is most consistent with an atypical AVNRT. She was given adenosine 12 mg IV by fast push at bedside while obtaining 12-lead rhythm strip. She converted to sinus rhythm and her dyspnea and chest discomfort resolved, as did the sagging ST-segment in the inferior leads. An ECG obtained after treatment is shown in Figure 5. Long-term prevention options include treatment with beta-blockers and possibly additional AV nodal agents, versus electrophysiology study / RF ablation.

This ECG is more likely to be accelerated junctional rhythm because the rate is < 120. Rates under 120 are unusual for AVNRT.
Steve Smith
The retrograde P wave is a bit far out from the end of the QRS to be an AVNRT. A fast-slow variant of AVNRT could result in an RP interval that long, but they are usually even longer, making the dysrhythmia a “long RP” tachycardia. That RP interval would be a lot more consistent with an AVRT, but like Dr. Smith, that would still be an awfully slow rate for an AVRT. An accelerated junctional rhythm is more likely.
Thanks for running this blog. It’s really great to see interest in the more advanced aspects of electrocardiography. Please keep up the good work!
Thank you for your kind words and your keen insight.