Medical knowledge, especially in toxicology, is built on experimentation and the preciously won experience over time treating the ingestions and poisonings of our patients. Ignoring these hard won lessons, and having additional patients repeat the suffering is a most pitiful waste. This has been the case with 2,4-dinitrophenol (DNP), which despite abundant evidence of its danger, continues to tempt because of its promise of weight loss.
Dinitrophenol Uses, Mechanism, and Effects
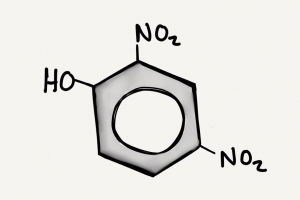
DNP is an industrial chemical widely used in manufacturing. Discovered in 1933, it incidentally was identified as promoting weight loss. As much as 2 lbs/week loss could be seen, which drove its use as a diet aid. DNP uncouples mitochondrial oxidative phosphorylation, interfering with the body’s ability to store energy as ATP. This leads to a dramatic increase in the metabolic rate, and hyperthermia to dissipate the heat. A secondary effect of depletion of ATP caused release of calcium from the sarcoplasmic reticulum and intractable muscle contractions which would generate additional thermal heat.
Unfortunately, its narrow therapeutic window, meant that even while taking recommended doses levels could become “toxic”, and severe side effects were seen including blindness (dinitrophenol cataracts) and death. Federal Food, Drug and Cosmetic Act of 1938 called the chemical “extremely dangerous and not fit for human consumption”. Popularity quickly waned and reported complications from the drug nearly disappeared.
Clinical Presentation and Treatment of Dinitrophenol Toxicity
Presenting symptoms usually include hyperthermia, nausea/vomiting, and diaphoresis. Severe neurological effects such as confusion, agitation, convulsions, and coma are common. The severe hyperthermia can be difficult to control, and patients die of hyperpyrexia and multiorgan failure and refractory shock.
Treatment is almost exclusively aggressive support cares.
- Fluid resuscitation
- Cooling for correction of hyperthermia
- Sedation
- Airway control
From a physiology mechanism, treatment with dantrolene would seem to offer benefit. Since dantrolene inhibits release of calcium from the sarcoplasmic reticulum, reduction in muscle contractions and heat generation should be possible. The threshold for treatment is set at 39-40°C by the U.K. National Poison Information Service (NPIS) guidelines.
Is DNP readily available?
Because of its reputation for weight loss the lessons of DNP have been forgotten or ignored, and the use of DNP is increasing along with occurrences of its toxic effects.
While DNP is certainly available over the internet, self-reported ingestions should be investigated closely. For example, on Amazon.com there are a large number of diet aids using the term DNP as a marketing ploy, without actually containing any DNP at all. True sources of DNP come from industrial or shadier sources.
Most of these diet aids, consist almost entirely very large amounts of caffeine which can generate symptoms similar to DNP.
- CNS: agitation, confusion, tremor, seizure
- Cardiovascular: tachycardia, palpitations
- Gastrointestinal: nausea, vomiting, diarrhea
However, the prominent feature of hyperthermia, muscle contractions, and severe lactic acidosis would not be seen in large caffeine ingestions and could be indicators of true DNP toxicity.
References
- Clinical features and treatment in patients with acute 2,4-dinitrophenol poisoning. Lu Y et al. J Zhejiang. University Science Biomed & Biotechnol 2011;12:189-192.
- 2,4-Dinitrophenol (DNP): A Weight Loss Agent with Significant Acute Toxicity and Risk of Death. Grundlingh J et al. J Med Toxicol 2011 July 8.
- Fatal 2,4-dinitrophenol poisoning…coming to a hospital near you. Siegmueller C, Narasimhaish R. Emerg Med J 2010 May 29.

My son overdosed on dip and died
I am terribly sorry for your loss. I hope we can make doctors and others aware of the danger of DNP.
My son also overdosed on DNP and died. ER doctors don’t seem to be well educated about this drug let alone how to treat patients who overdose on it.
My sister overdosed on it last week and died, th A&E did nothing but ‘keep and eye on her’ until the point she cardiac arested but by then it was too late! The doctor then told my mum after – sorry we didn’t know what to do , we’ve never seen this before!
A friend’s wife arrested and died last week as a result of taking too much of this substance. It seems (like the others who have commented) the ER here was not prepared to deal effectively with this problem. Sad on all levels. So very sad.
Sorry for everyones loss. Eiggez, this was so recent. When you said “here” was that in the US?
Sorry about all these losses. eiggez, use among females worldwide seems to be increasing. I’m assuming your friends wife was located in the US? So sorry!
i am actually watching and episode of Untold Story of the ER and DNP is actually on there. There is no known antidote for DNP that is why people are dieing on this weight loss product because your temp keeps rising and you can’t get it to come down at all nothing helps no cold compresses or fans water nothing and it is sad i think that ER Doctors need to be educated in this. In 1933 the FDA banned DNP the side effects are breathing decreased temp keeps rising and your o2 stats go down. there is only one thing that could help but not 100 percent and that drug is called dantrolene which is a muscle relaxant and even that is questionable and not 100% and you can even still die even if given dantrolene but like i said no know antidote for DNP exist. The patient on the episode of Untold Stories Of The ER was given 2 rounds of dantrolene and she survived. For those people on this page who died I am so sorry for there family and their friends.